Abstract
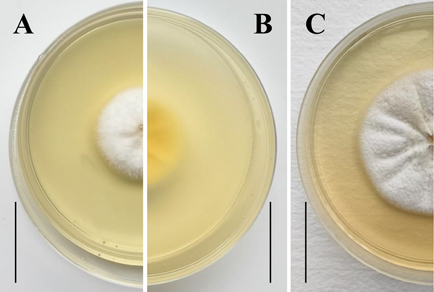
The family Stachybotryaceae (Hypocreales) has recently undergone substantial taxonomic revision, with multilocus phylogenetics revealing numerous cryptic lineages. During a survey of soil fungi in Guizhou Province, China, a myrothecium-like fungus was isolated and is here described as Myxospora guizhouensis sp. nov. based on morphological and molecular evidence. The new species is characterized by solitary, hyaline, flask-shaped to cylindrical conidiogenous cells arising directly from vegetative hyphae, and by solitary, acrogenous conidia that are globose to limoniform, hyaline, aseptate, thin-walled, and smooth. Phylogenetic analyses of a combined ITS, nrLSU, tef-1α, rpb2, tub2, and cmdA dataset using maximum likelihood and Bayesian inference resolve M. guizhouensis as a distinct, well-supported lineage within Myxospora, closely related to M. crassiseta. Both morphological comparisons and molecular phylogenetic data support the recognition of M. guizhouensis as a new species, representing the first Myxospora species reported from soil in China.
References
- Carbone, I. & Kohn, L.M. (1999) A method for designing primer sets for speciation studies in filamentous ascomycetes. Mycologia 91: 553–556. https://doi.org/10.1080/00275514.1999.12061051
- Chen,Y.P., Lu, Z.H., Faraj, T.K. & Maharachchikumbura, S.S.N. (2023) Myxospora poaceicola sp. nov. (Stachybotryaceae, Hypocreales), a novel myrothecium-like fungus from Digitaria sanguinalis (Poaceae). Phytotaxa 625: 280–288. https://doi.org/10.11646/phytotaxa.625.3.4
- Chen, H., Bibi, S., Tao, L., Shen, X.C., Zhao, J., Sun, Y.M., Li, Q.R., Tang, D.X. & Wang, Y. (2025) Papiliomyces sinensis (Clavicipitaceae) and Paraisaria pseudoarcta (Ophiocordycipitaceae), two new species parasitizing Lepidopteran insects from southwestern China. MycoKeys 117: 353–374. https://doi.org/10.3897/mycokeys.117.150376
- Cheng, K.W., Yang, J., Srimongkol, P., Stadler, M., Karnchanatat, A. & Ariyawansa, H.A. (2025) Fungal frontiers in toxic terrain: revealing culturable fungal communities in serpentine paddy fields of Taiwan. IMA Fungus 16: e155308. https://doi.org/10.3897/imafungus.16.155308
- Crous, P.W., Shivas, R.G., Quaedvlieg, W., van der Bank, M., Zhang, Y., Summerell, B.A., Guarro, J., Wingfield, M.J., Wood, A.R., Alfenas, A.C., Braun,U., Cano-Lira, J.F., García, D., Marin-Felix, Y., Alvarado, P., Andrade, J.P., Armengol, J., Assefa, A., den Breeÿen, A., Camele,I., Cheewangkoon, R., De Souza, J.T., Duong, T.A., Esteve-Raventós, F., Fournier, J., Frisullo, S., García-Jiménez, J., Gardiennet, A., Gené, J., Hernández-Restrepo, M., Hirooka, Y., Hospenthal, D.R., King, A., Lechat, C., Lombard, L., Mang, S.M., Marbach, P.A.S., Marincowitz, S., Montaño-Mata, N.J., Moreno, G., Perez, C.A., Pérez Sierra, A.M., Robertson, J.L., Roux, J., Rubio, E., Schumacher, R.K., Stchigel, A.M., Sutton, D.A., Tan, Y.P., Thompson, E.H., Van der linde, E., Walker, A.K., Walker, D.M., Wickes, B.L., Wong, P.T.W. & Groenewald, J.Z. (2014) Fungal Planet description sheets: 214–280. Persoonia 32: 184–306. https://doi.org/10.3767/003158514X682395
- Darriba, D., Taboada, G.L., Doallo, R. & Posada, D. (2012) jModelTest 2: more models, new heuristics and parallel computing. Nature Methods 9: 772. https://doi.org/10.1038/nmeth.2109
- Groenewald, J.Z., Nakashima, C., Nishikawa, J., Shin, H.D., Park, J.H., Jama, A.N., Groenewald, M. & Crous, P.W. (2013) Species concepts in Cercospora: spotting the weeds among the roses. Studies in Mycology 75: 115–170. https://doi.org/10.3114/sim0012
- He, S.C., Thiyagaraja, V., Bhunjun, C.S., Jayawardena, R.S., Chomnunti, P., Al-Otibi, F., Zhao, Y.W., Zhao, Q. & Hyde, K.D. (2024) A new Myxospora (Hypocreales, Sordariomycetes) species from the Tibetan Plateau, China. New Zealand Journal of Botany 63: 1193–1209. https://doi.org/10.1080/0028825X.2024.2438411
- Hoang, D.T., Chernomor, O., von Haeseler, A., Minh, B.Q. & Vinh, L.S. (2018) UFBoot2: improving the ultrafast bootstrap approximation. Molecular Biology and Evolution 35: 518–522. https://doi.org/10.1093/molbev/msx281
- Hyde, K.D., Dong, Y., Phookamsak, R., Jeewon, R., Bhat, D.J., Jones, E.B.G., Liu, N.G., Abeywickrama, P.D., Mapook, A., Wei, D., Perera, R.H., Manawasinghe, I.S., Pem, D., Bundhun, D., Karunarathna, A., Ekanayaka, A.H., Bao, D.F., Li, J., Samarakoon, M.C., Chaiwan, N., Lin, C.G., Phutthacharoen, K., Zhang, S.N., Senanayake, I.C., Goonasekara, I.D., Thambugala, K.M., Phukhamsakda, C., Tennakoon, D.S., Jiang, H.B., Yang, J., Zeng, M., Huanraluek, N., Liu, J.K., Wijesinghe, S.N., Tian, Q., Tibpromma, S., Brahmanage, R.S., Boonmee, S., Huang, S.K., Thiyagaraja, V., Lu, Y.Z., Jayawardena, R.S., Dong, W., Yang, E.F., Singh, S.K., Singh, S.M., Rana, S., Lad, S.S., Anand, G., Devadatha, B., Niranjan, M., Sarma, V.V., Liimatainen, K., Aguirre-Hudson, B., Niskanen, T., Overall, A., Alvarenga, R.L.M., Gibertoni, T.B., Pfliegler, W.P., Horváth, E., Imre, A., Alves, A.L., Santos, A.C.S., Tiago, P.V., Bulgakov, T.S., Wanasinghe, D.N., Bahkali, A.H., Doilom, M., Elgorban, A.M., Maharachchikumbura, S.S.N., Rajeshkumar, K.C., Haelewaters, D., Mortimer, P.E., Zhao, Q., Lumyong, S., Xu, J. & Sheng, J. (2025) Fungal diversity notes 2017–2122: taxonomic and phylogenetic contributions to freshwater fungi and other fungal taxa. Fungal Diversity 134: 185–459. https://doi.org/10.1007/s13225-025-00560-3
- Hyde, K.D., Norphanphoun, C., Maharachchikumbura, S.S.N., Bhat, D.J., Jones, E.B.G., Bundhun, D., Chen, Y.J., Bao, D.F., Boonmee, S., Calabon, M.S., Chaiwan, N., Chethana, K.W.T., Dai, D.Q., Dayarathne, M.C., Devadatha, B., Dissanayake, A.J., Dissanayake, L.S., Doilom, M., Dong, W., Fan, X.L., Goonasekara, I.D., Hongsanan, S., Huang, S.K., Jayawardena, R.S., Jeewon, R., Karunarathna, A., Konta, S., Kumar, V., Lin, C.G., Liu, J.K., Liu, N.G., Luangsa-ard, J., Lumyong, S., Luo, Z.L., Marasinghe, D.S., McKenzie, E.H.C., Niego, A.G.T., Niranjan, M., Perera, R.H., Phukhamsakda, C., Rathnayaka, A.R., Samarakoon, M.C., Samarakoon, S.M.B.C., Sarma, V.V., Senanayake, I.C., Shang, Q.J., Stadler, M., Tibpromma, S., Wanasinghe, D.N., Wei, D.P., Wijayawardene, N.N., Xiao,Y.P., Yang, J., Zeng, X.Y., Zhang, S.N. & Xiang, M.M. (2020) Refined families of Sordariomycetes. Mycosphere 11: 305–1059. https://doi.org/10.5943/mycosphere/11/1/7
- Kalyaanamoorthy, S., Minh, B.Q., Wong, T.K.F., von Haeseler, A. & Jermiin, L.S. (2017) ModelFinder: fast model selection for accurate phylogenetic estimates. Nature Methods 14: 587–589. https://doi.org/10.1038/nmeth.4285
- Liang, J.M., Li, G.S., Zhou, S.Y., Zhao, M.Q. & Cai, L. (2019) Myrothecium-like new species from turfgrasses and associated rhizosphere. MycoKeys 51: 29–53. https://doi.org/10.3897/mycokeys.51.31957
- Liu, Y.J., Whelen, S. & Hall, B.D. (1999) Phylogenetic relationships among ascomycetes: evidence from an RNA polymerase II subunit. Molecular Biology and Evolution 16: 1799–1808. https://doi.org/10.1093/oxfordjournals.molbev.a026092
- Lombard, L., Houbraken, J., Decock, C., Samson, R.A., Meijer, M., Réblová, M., Groenewald, J.Z. & Crous, P.W. (2016) Generic hyper-diversity in Stachybotryaceae. Persoonia 36: 156–246. https://doi.org/10.3767/003158516X691582
- O’Donnell, K. & Cigelnik, E. (1997) Two divergent intragenomic rDNA ITS2 types within a monophyletic lineage of Fusarium are nonorthologous. Molecular Phylogenetics and Evolution 7: 103–116. https://doi.org/10.1006/mpev.1996.0376
- O’Donnell, K., Kistler, H.C., Cigelnik, E. & Ploetz, R.C. (1998) Multiple evolutionary origins of the fungus causing Panama disease of banana: concordant evidence from nuclear and mitochondrial gene genealogies. Proceedings of the National Academy of Sciences of the United States of America 95: 2044–2049. https://doi.org/10.1073/pnas.95.5.2044
- Phukhamsakda, C., McKenzie, E.H.C., Phillips, A.J.L., Jones, E.B.G., Bhat, D.J., Stadler, M., Bhunjun, C.S., Wanasinghe, D.N., Thongbai, B., Camporesi, E., Ertz, D., Jayawardena, R.S., Perera, R.H., Ekanayake, A.H., Tibpromma, S., Doilom, M. & Hyde, K.D. (2020) Microfungi associated with Clematis (Ranunculaceae) with an integrated approach to delimiting species boundaries. Fungal Diversity 102: 1–203. https://doi.org/10.1007/s13225-020-00448-4
- Ronquist, F., Teslenko, M., van der Mark, P., Ayres, D.L., Darling, A., Höhna, S., Larget, B., Liu, L., Suchard, M.A. & Huelsenbeck, J.P. (2012) MrBayes 3.2: efficient Bayesian phylogenetic inference and model choice across a large model space. Systematic Biology 61: 539–542. https://doi.org/10.1093/sysbio/sys029
- Samarakoon, B.C., Wanasinghe, D.N., Bhat, J. & Chomnunti, P. (2022) Taxonomy and phylogeny of Smaragdiniseta musae sp. nov. and Albifimbria verrucaria (Hypocreales, Stachybotryaceae) on Musa from Thailand. Biodiversity Data Journal 10: e89360. https://doi.org/10.3897/BDJ.10.e89360
- Stamatakis, A., Hoover, P. & Rougemont, J. (2008) A rapid bootstrap algorithm for the RAxML Web servers. Systematic Biology 57: 758–771. https://doi.org/10.1080/10635150802429642
- Tamura, K., Stecher, G., Peterson, D., Filipski, A. & Kumar, S. (2013) MEGA6: molecular evolutionary genetics analysis version 6.0. Molecular Biology and Evolution 30: 2725–2729. https://doi.org/10.1093/molbev/mst197
- Vieto,S., Escudero-Leyva, E., Avendaño, R., Rechnitzer, N., Barrantes-Madrigal, M.D., Conejo-Barboza, G., Herrera-Sancho, O.A., Chaverri, P. & Chavarría, M. (2022) Biodeterioration and cellulolytic activity by fungi isolated from a nineteenth-century painting at the National Theatre of Costa Rica. Fungal Biology 126: 101–112. https://doi.org/10.1016/j.funbio.2021.11.001
- Vilgalys, R. & Hester, M. (1990) Rapid genetic identification and mapping of enzymatically amplified ribosomal DNA from several Cryptococcus species. Journal of Bacteriology 172: 4238–4246. https://doi.org/10.1128/JB.172.8.4238-4246.1990
- Wang, Y., Hyde, K.D., McKenzie, E.H.C., Jiang, Y.L., Li, D.W. & Zhao, D.G. (2015) Overview of Stachybotrys (Memnoniella) and current species status. Fungal Diversity 71: 17–83. https://doi.org/10.1007/s13225-014-0319-0
- Wang, Y., Tang, D.X., Chen, H., Li, Q.R., Loinheuang, C. & Shen, X.C. (2025) Phylogenetic evidence reveals a close relationship between Amphichorda and Ovicillium in Bionectriaceae (Hypocreales). MycoKeys 117: 337–352. https://doi.org/10.3897/mycokeys.117.151366
- White, T.J., Bruns, T., Lee, S.B. & Taylor, J.W. (1990) Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In: Innis,M.A., Gelfand,D.H., Sninsky,J.J. &White,T.J. (Eds.) PCR Protocols: A Guide to Methods and Applications. Academic Press, San Diego, pp. 315–322. https://doi.org/10.1016/B978-0-12-372180-8.50042-1
- Zhang, J.Y., Hyde, K.D., Bao, D.F., Hongsanan, S., Liu, Y.X., Kang, J.C., Luo, Z.L., Liu, J.K. & Lu,Y.Z. (2025) A worldwide checklist and morpho-molecular systematics of fungi associated with pteridophytes. Fungal Diversity 132: 151–423. https://doi.org/10.1007/s13225-025-00554-1