Abstract
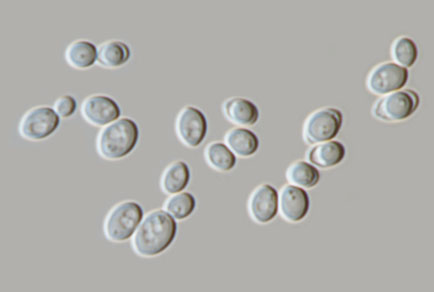
During a survey of leaf litter associated yeasts, a novel species Monosporozyma xishuangbannaensis was isolated from the leaves of Ficus religiosa in Xishuangbanna, Yunnan Province, China. The identification of the species was based on molecular data, morphological, physiological, and biochemical properties. Maximum likelihood (ML) and Bayesian inference (BI) analyses of the internal transcribed spacer (ITS) region and the D1/D2 domains of the large subunit (LSU) rDNA sequence dataset were used to confirm placement of the isolate. The phylogenetic analysis based on the D1/D2 domains of LSU sequences showed that the two strains represented a novel species in Monosporozyma. Maximum likelihood and Bayesian inference analyses revealed that Monosporozyma xishuangbannaensis formed a distinct clade with the ex-type isolate of M. unispora (CBS 398) with 97% ML and 1.00 PP statistical support, but they have 1.23% difference in the D1/D2 domains of LSU and 1.87% difference in the ITS region. Biochemical and physiological characteristics showed differences in some carbon assimilation and nitrogen profiles compared to M. unispora.
References
- Aerts, R. (1997) Climate, leaf litter chemistry and leaf litter decomposition in terrestrial ecosystems: a triangular relationship. Oikos 79: 439–449. https://doi.org/10.2307/3546886
- Baldrian, P. (2017) Microbial activity and the dynamics of ecosystem processes in forest soils. Current Opinion in Microbiology 37: 128–134. https://doi.org/10.1016/j.mib.2017.06.008
- Bani, A., Pioli, S., Ventura, M., Panzacchi, P., Borruso, L., Tognetti, R., Tonon, G. & Brusetti, L. (2018) The role of microbial community in the decomposition of leaf litter and deadwood. Applied Soil Ecology 126: 75–84. https://doi.org/10.1016/j.apsoil.2018.02.017
- Berg, B. & McClaugherty, C. (2008) Chemical constituents as rate-regulating: initial variation and changes during decomposition. In: Berg, B. & McClaugherty, C. (2nd Ed.) Plant Litter: Decomposition, Humus Formation, Carbon Sequestration. Springer, Berlin, Heidelberg, pp. 115–148. https://doi.org/10.1007/978-3-540-74923-3_6
- Boekhout, T., Bai, F.Y., Daniel, H.M., Groenewald, M., Robert, V., Tan, C.S. & Yurkov, A. (2025) The Yeasts Trust Database. Uppsalaan 8, 3534CT Utrecht, The Netherlands. Available from: https://theyeasts.org/ (accessed 7 August 2025)
- Botha, A. (2011) The importance and ecology of yeasts in soil. Soil Biology and Biochemistry 43 (1): 1–8. https://doi.org/10.1016/j.soilbio.2010.10.001
- Cadete, R.M., Lopes, M.R. & Rosa, C.A. (2017) Yeasts associated with decomposing plant material and rotting wood. In: Buzzini, P., Lachance, M.A. & Yurkov, A. (Eds.) Yeasts in Natural Ecosystems: Diversity. Springer, Cham, pp. 265–292. https://doi.org/10.1007/978-3-319-62683-3_9
- Dabassa Koricha, A., Han, D.Y., Bacha, K. & Bai, F.Y. (2019) Occurrence and molecular identification of wild yeasts from Jimma Zone, South West Ethiopia. Microorganisms 7 (12): 633. https://doi.org/10.3390/microorganisms7120633
- Darriba, D., Taboada, G.L., Doallo, R. & Posada, D. (2012) jModelTest 2: more models, new heuristics and high-performance computing. Nature Methods 9 (8): 772. https://doi.org/10.1038/nmeth.2109
- Gonçalves Jr, J.F., França, J.S., Medeiros, A.O., Rosa, C.A. & Callisto, M. (2006) Leaf breakdown in a tropical stream. International Review of Hydrobiology 91 (2): 164–177. https://doi.org/10.1002/iroh.200510826
- Gouliamova, D. & Dimitrov, R. (2020) Kazachstania chrysolinae and Kazachstania bozae two new yeast species of the genus Kazachstania. Transfer of four Kazachstania species to Grigorovia gen. nov. as new combinations. Comptes Rendus de l’Académie Bulgare des Sciences 73 (1): 48–57. https://doi.org/10.7546/CRABS.2020.01.06
- Hall, T.A. (1999) BioEdit: a user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucleic Acids Symposium Series 41 (41): 95–98.
- Huelsenbeck, J.P., Ronquist, F., Nielsen, R. & Bollback, J.P. (2001) Bayesian inference of phylogeny and its impact on evolutionary biology. Science 294: 2310–2314. https://doi.org/10.1126/science.1065889
- Hyde, K.D., Noorabadi, M.T., Thiyagaraja, V., He, M.Q., Johnston, P.R., Wijesinghe, S.N., Armand, A., Biketova, A.Y., Chethana, K.W.T., Erdoğdu, M., Ge, Z.W., Groenewald, J.Z., Hongsanan, S., Kušan, I., Leontyev, D.V., Li, D.W., Lin, C.G., Liu, N.G., Maharachchikumbura, S.S.N., Matočec, N., May, T.W., McKenzie, E.H.C., Mešić, A., Perera, R.H., Phukhamsakda, C., Piątek, M., Samarakoon, M.C., Selcuk, F., Senanayake, I.C., Tanney, J.B., Tian, Q., Vizzini, A., Wanasinghe, D.N., Wannasawang, N., Wijayawardene, N.N., Zhao, R.L., Abdel-Wahab, M.A., Abdollahzadeh, J., Abeywickrama, P.D., Abhinav, Absalan, S., Acharya, K., Afshari, N., Afshan, N.S., Afzalinia, S., Ahmadpour, S.A., Akulov, O., Alizadeh, A., Alizadeh, M., Al-Sadi, A.M., Alves, A., Alves, V.C.S., Alves-Silva, G., Antonín, V., Aouali, S., Aptroot, A., Apurillo, C.C.S., Arias, R.M., Asgari, B., Asghari, R., Assis, D.M.A., Assyov, B., Atienza, V., Aumentado, H.D.R., Avasthi, S., Azevedo, E., Bakhshi, M., Bao, D.F., Baral, H.O., Barata, M., Barbosa, K.D., Barbosa, R.N., Barbosa, F.R., Baroncelli, R., Barreto, G.G., Baschien, C., Bennett, R.M., Bera, I., Bezerra, J.D.P., Bhunjun, C.S., Bianchinotti, M.V., Błaszkowski, J., Boekhout, T., Bonito, G.M., Boonmee, S., Boonyuen, N., Bortnikov, F.M., Bregant, C., Bundhun, D., Burgaud, G., Buyck, B., Caeiro, M.F., Cabarroi-Hernández, M., Cai, M.F., Cai, L., Calabon, M.S., Calaça, F.J.S., Callalli, M., Câmara, M.P.S., Cano-Lira, J., Cao, B., Carlavilla, J.R., Carvalho, A., Carvalho, T.G., Castañeda-Ruiz, R.F., Catania, M.D.V., Cazabonne, J., Cedeño-Sanchez, M., Chaharmiri-Dokhaharani, S., Chaiwan, N., Chakraborty, N., Cheewankoon, R., Chen, C., Chen, J., Chen, Q., Chen, Y.P., Chinaglia, S., Coelho-Nascimento, C.C., Coleine, C., CostaRezende, D.H., Cortés-Pérez, A., Crouch, J.A., Crous, P.W., Cruz, R.H.S.F., Czachura, P., Damm, U., Darmostuk, V., Daroodi, Z., Das, K., Das, K., Davoodian, N., Davydov, E.A., da Silva, G.A., da Silva, I.R., da Silva, R.M.F., da Silva Santos, A.C., Dai, D.Q., Dai, Y.C., de Groot, D., De Kesel, A., De Lange, R., de Medeiros, E.V., de Souza, C.F.A., de Souza, F.A., dela Cruz, T.E.E., Decock, C., Delgado, G., Denchev, C.M., Denchev, T.T., Deng, Y.L., Dentinger, B.T.M., Devadatha, B., Dianese, J.C., Dima, B., Doilom, M., Dissanayake, A.J., Dissanayake, D.M.L.S., Dissanayake, L.S., Diniz, A.G., Dolatabadi, S., Dong, J.H., Dong, W., Dong, Z.Y., Drechsler-Santos, E.R., Druzhinina, I.S., Du, T.Y., Dubey, M.K., Dutta, A.K., Elliott, T.F., Elshahed, M.S., Egidi, E., Eisvand, P., Fan, L., Fan, X., Fan, X.L., Fedosova, A.G., Ferro, L.O., Fiuza, P.O., Flakus, A., Fonseca, E.O.W., Fryar, S.C., Gabaldón, T., Gajanayake, A.J., Gannibal, P.B., Gao, F., García-Sánchez, D., García-Sandoval, R., Garrido-Benavent, I., Garzoli, L., Gasca-Pineda, J., Gautam, A.K., Gené, J., Ghobad-Nejhad, M., Ghosh, A., Giachini, A.J., Gibertoni, T.B., Gentekaki, E., Gmoshinskiy, V.I., Góes-Neto, A., Gomdola, D., Gorjón, S.P., Goto, B.T., Granados-Montero, M.M., Griffith, G.W., Groenewald, M., Grossart, H.P., Gu, Z.R., Gueidan, C., Gunarathne, A., Gunaseelan, S., Guo, S.L., Gusmão, L.F.P., Gutierrez, A.C., Guzmán-Dávalos, L., Haelewaters, D., Haituk, H., Halling, R.E., He, S.C., Heredia, G., Hernández-Restrepo,M., Hosoya, T., Hoog, S.D., Horak, E., Hou, C.L., Houbraken, J., Htet, Z.H., Huang, S.K., Huang, W.J., Hurdeal, V.G., Hustad, V.P., Inácio, C.A., Janik, P., Jayalal, R.G.U., Jayasiri, S.C., Jayawardena, R.S., Jeewon, R., Jerônimo, G.H., Jin, J., Jones, E.B.G., Joshi, Y., Jurjević, Ž., Justo, A., Kakishima, M., Kaliyaperumal, M., Kang, G.P., Kang, J.C., Karimi, O., Karunarathna, S.C., Karpov, S.A., Kezo, K., Khalid, A.N., Khan, M.K., Khuna, S., Khyaju, S., Kirchmair, M., Klawonn, I., Kraisitudomsook, N., Kukwa, M., Kularathnage, N.D., Kumar, S., Lachance, M.A., Lado, C., Latha, K.P.D., Lee, H.B., Leonardi, M., Lestari, A.S., Li, C., Li, H., Li, J., Li, Q., Li, Y., Li, Y.C., Li, Y.X., Liao, C.F., Lima, J.L.R., Lima, J.M.S., Lima, N.B., Lin, L., Linaldeddu, B.T., Linn, M.M., Liu, F., Liu, J.K., Liu, J.W., Liu, S., Liu, S.L., Liu, X.F., Liu, X.Y., Longcore, J.E., Luangharn, T., Luangsa-ard, J.J., Lu, L., Lu, Y.Z., Lumbsch, H.T., Luo, L., Luo, M., Luo, Z.L., Ma, J., Madagammana, A.D., Madhushan, A., Madrid, H., Magurno, F., Magyar, D., Mahadevakumar, S., Malosso, E., Malysh, J.M., Mamarabadi, M., Manawasinghe, I.S., Manfrino, R.G., Manimohan, P., Mao, N., Mapook, A., Marchese, P., Marasinghe, D.S., Mardones, M., Marin-Felix, Y., Masigol, H., Mehrabi, M., Mehrabi-Koushki, M., Meiras-Ottoni, A., Melo, R.F.R., Mendes-Alvarenga, R.L., Mendieta, S., Meng, Q.F., Menkis, A., Menolli Jr, N., Mikšík, M., Miller, S.L., Moncada, B., Moncalvo, J.M., Monteiro, J.S., Monteiro, M., Mora-Montes, H.M., Moroz, E.L., Moura, J.C., Muhammad, U., Mukhopadhyay, S., Nagy, G.L., Najam ul Sehar, A., Najafiniya, M., Nanayakkara, C.M., Naseer, A., Nascimento, E.C.R., Nascimento, S.S., Neuhauser, S., Neves, M.A., Niazi, A.R., Yong, N., Nilsson, R.H., Nogueira, P.T.S., Novozhilov, Y.K., Noordeloos, M., Norphanphoun, C., Nuñez-Otaño, N., O’Donnell, R.P., Oehl, F., Oliveira, J.A., Oliveira Junior, I., Oliveira, N.V.L., Oliveira, P.H.F., Orihara, T., Oset, M., Pang, K.L., Papp, V., Pathirana, L.S., Peintner, U., Pem, D., Pereira, O.L., Pérez-Moreno, J., Pérez-Ortega, S., Péter, G., Pires-Zottarelli, C.L.A., Phonemany, M., Phongeun, S., Pošta, A., Prazeres, J.F.S.A., Quan, Y., Quandt, C.A., Queiroz, M.B., Radek, R., Rahnama, K., Raj, K.N.A., Rajeshkumar, K.C., Soumyadeep, R., Ralaiveloarisoa, A.B., Rämä, T., Ramírez-Cruz, V., Rambold, G., Rathnayaka, A.R., Raza, M., Ren, G.C., Rinaldi, A.C., Rivas-Ferreiro, M., Robledo, G.L., Ronikier, A., Rossi, W., Rusevska, K., Ryberg, M., Safi, A., Salimi, F., Salvador-Montoya, C.A., Samant, B., Samaradiwakara, N.P., Sánchez-Castro, I., Sandoval-Denis, M., Santiago, A.L.C.M.A., Santos, A.C.D.S., Santos, L.A. dos, Sarma, V.V., Sarwar, S., Savchenko, A., Savchenko, K., Saxena, R.K., Schoutteten, N., Selbmann, L., Ševčíková, H., Sharma, A., Shen, H.W., Shen, Y.M., Shu, Y.X., Silva, H.F., Silva-Filho, A.G.S., Silva, V.S.H., Simmons, D.R., Singh, R., Sir, E.B., Sohrabi, M., Souza, F.A., Souza-Motta, C.M., Sriindrasutdhi, V., Sruthi, O.P., Stadler, M., Stemler, J., Stephenson, S.L., Stoyneva-Gaertner, M.P., Strassert, J.F.H., Stryjak-Bogacka, M., Su, H., Sun, Y.R., Svantesson, S., Sysouphanthong, P., Takamatsu, S., Tan, T.H., Tanaka, K., Tang, C., Tang, X., Taylor, J.E., Taylor, P.W.J., Tennakoon, D.S., Thakshila, S.A.D., Thambugala, K.M., Thamodini, G.K., Thilanga, D., Thines, M., Tiago, P.V., Tian, X.G., Tian, W.H., Tibpromma, S., Tkalčec, Z., Tokarev, Y.S., Tomšovský, M., Torruella, G., Tsurykau, A., Udayanga, D., Ulukapı, M., Untereiner, W.A., Usman, M., Uzunov, B.A., Vadthanarat, S., Valenzuela, R., Van den Wyngaert, S., Van Vooren, N., Velez, P., Verma, R.K., Vieira, L.C., Vieira, W.A.S., Vinzelj, J.M., Tang, A.M.C., Walker, A., Walker, A.K., Wang, Q.M., Wang, Y., Wang, X.Y., Wang, Z.Y., Wannathes, N., Wartchow, F., Weerakoon, G., Wei, D.P., Wei, X., White, J.F., Wijesundara, D.S.A., Wisitrassameewong, K., Worobiec, G., Wu, H.X., Wu, N., Xiong, Y.R., Xu, B., Xu,J.P., Xu, R., Xu, R.F., Xu, R.J., Yadav, S., Yakovchenko, L.S., Yang, H.D., Yang, X., Yang, Y.H., Yang, Y., Yang, Y.Y., Yoshioka, R., Youssef, N.H., Yu, F.M., Yu, Z.F., Yuan, L.L., Yuan, Q., Zabin, D.A., Zamora, J.C., Zapata, C.V., Zare, R., Zeng, M., Zeng, X.Y., Zhang, J.F., Zhang, J.Y., Zhang, S., Zhang, X.C., Zhao, C.L., Zhao, H., Zhao, Q., Zhao, H., Zhao, H.J., Zhou, H.M., Zhu, X.Y., Zmitrovich, I.V., Zucconi, L. & Zvyagina, E. (2024) The 2024 outline of fungi and fungus-like taxa. Mycosphere 15 (1): 5146–6239. https://doi.org/10.5943/mycosphere/15/1/25
- Index Fungorum (2025) Available from: http://www.indexfungorum.org/Names/Names.asp (accessed 22 October 2025)
- James, S.A., Carvajal Barriga, E.J., Portero Barahona, P., Nueno-Palop, C., Cross, K., Bond, C.J. & Roberts, I.N. (2015) Kazachstania yasuniensis sp. nov., an ascomycetous yeast species found in mainland Ecuador and on the Galápagos. International Journal of Systematic and Evolutionary Microbiology 65: 1304–1309. https://doi.org/10.1099/ijs.0.000102
- Jayasiri, S.C., Hyde, K.D., Ariyawansa, H.A., Bhat, D.J., Buyck, B., Cai, L., Dai, Y.-C., Abd-Elsalam, K.A., Ertz, D., Hidayat, I., Jeewon, R., Jones, E.B.G., Bahkali, A.H., Karunarathna, S.C., Liu, J.-K., Luangsa-ard, J.J., Lumbsch, H.T., Maharachchikumbura, S.S.N., McKenzie, E.H.C., Moncalvo, J.-M., Ghobad-Nejhad, M., Nilsson, H., Pang, K.-L., Pereira, O.L., Phillips, A.J.L., Raspé, O., Rollins, A.W., Romero, A.I., Etayo, J., Selçuk, F., Stephenson, S.L., Suetrong, S., Taylor, J.E., Tsui, C.K.M., Vizzini, A., Abdel-Wahab, M.A., Wen, T.-C., Boonmee, S., Dai, D.Q., Daranagama, D.A., Dissanayake, A.J., Ekanayaka, A.H., Fryar, S.C., Hongsanan, S., Jayawardena, R.S., Li, W.-J., Perera, R.H., Phookamsak, R., de Silva, N.I., Thambugala, K.M., Tian, Q., Wijayawardene, N.N., Zhao, R.-L. & Promputtha, I. (2015) The Faces of Fungi database: fungal names linked with morphology, phylogeny and human impacts. Fungal Diversity 74: 3–18. https://doi.org/10.1007/s13225-015-0351-8
- Junyapate, K., Jindamorakot, S. & Limtong, S. (2014) Yamadazyma ubonensis fa, sp. nov., a novel xylitol-producing yeast species isolated in Thailand. Antonie van Leeuwenhoek 105 (3): 471–480. https://doi.org/10.1007/s10482-013-0098-8
- Katoh, K., Rozewicki, J. & Yamada, K.D. (2019) MAFFT online service: multiple sequence alignment, interactive sequence choice and visualization. Briefings in Bioinformatics 20 (4): 1160–1166. https://doi.org/10.1093/bib/bbx108
- Khan, Z.U., Randhawa, H.S., Kowshik, T., Chowdhary, A. & Chandy, R. (2007) Antifungal susceptibility of Cryptococcus neoformans and Cryptococcus gattii isolates from decayed wood of trunk hollows of Ficus religiosa and Syzygium cumini trees in north-western India. Journal of Antimicrobial Chemotherapy 60 (2): 312–316. https://doi.org/10.1093/jac/dkm192
- Khan, Z.U., Ahmad, S., Hagen, F., Fell, J.W., Kowshik, T., Chandy, R. & Boekhout, T. (2010) Cryptococcus randhawai sp. nov., a novel anamorphic basidiomycetous yeast isolated from tree trunk hollow of Ficus religiosa (peepal tree) from New Delhi, India. Antonie van Leeuwenhoek 97 (3): 253–259. https://doi.org/10.1007/s10482-009-9406-8
- Kowallik, V. & Greig, D. (2016) A systematic forest survey showing an association of Saccharomyces paradoxus with oak leaf litter. Environmental Microbiology Reports 8 (5): 833–841. https://doi.org/10.1111/1758-2229.12446
- Kumar, S., Stecher, G., Suleski, M., Sanderford, M., Sharma, S. & Tamura, K. (2024) MEGA12: Molecular Evolutionary Genetic Analysis version 12 for adaptive and green computing. Molecular Biology and Evolution 41 (12): 263. https://doi.org/10.1093/molbev/msae263
- Kurtzman, C.P. & Robnett, C.J. (1998) Identification and phylogeny of ascomycetous yeasts from analysis of nuclear large subunit (26S) ribosomal DNA partial sequences. Antonie van Leeuwenhoek 73 (4): 331–371. https://doi.org/10.1023/A:1001761008817
- Kurtzman, C.P., Fell, J.W., Boekhout, T. & Robert, V. (2011) Methods for isolation, phenotypic characterization and maintenance of yeasts. In: Kurtzman, C.P., Fell, J.W. & Boekhout, T. (5th Ed.) The Yeasts. Elsevier, Amsterdam, pp. 87–110. https://doi.org/10.1016/B978-0-444-52149-1.00007-0
- Liang, K. (2024) The role of leaf litter in forest soil fertility and microbial diversity. Molecular Soil Biology 15 (3): 99–108. https://doi.org/10.5376/msb.2024.15.0011
- Limtong, S., Yongmanitchai, W., Tun, M.M., Kawasaki, H. & Seki, T. (2007) Kazachstania siamensis sp. nov., an ascomycetous yeast species from forest soil in Thailand. International Journal of Systematic and Evolutionary Microbiology 57 (2): 419–422. https://doi.org/10.1099/ijs.0.64539-0
- Liu, X.Z., Wang, Q.M., Göker, M., Groenewald, M., Kachalkin, A.V., Lumbsch, H.T., Millanes, A.M., Wedin, M., Yurkov, A.M., Boekhout, T. & Bai, F.Y. (2015) Towards an integrated phylogenetic classification of the Tremellomycetes. Studies in Mycology 81 (1): 85–147. https://doi.org/10.1016/j.simyco.2015.12.001
- Liu, F., Hu, Z.D., Yurkov, A., Chen, X.H., Bao, W.J., Ma, Q., Zhao, W.N., Pan, S., Zhao, X.M., Liu, J.H., Wang, Q.M. & Boekhout, T. (2024) Saccharomycetaceae: delineation of fungal genera based on phylogenomic analyses, genomic relatedness indices and genomics-based synapomorphies. Persoonia 52 (1): 1–21. https://doi.org/10.3767/persoonia.2024.52.01
- Lu, H.Z., Cai, Y., Wu, Z.W., Jia, J.H. & Bai, F.Y. (2004) Kazachstania aerobia sp. nov., an ascomycetous yeast species from aerobically deteriorating corn silage. International Journal of Systematic and Evolutionary Microbiology 54 (6): 2431–2435. https://doi.org/10.1099/ijs.0.63257-0
- Maksimova, I.A. & Chernov, I.Y. (2004) Community structure of yeast fungi in forest biogeocenoses. Microbiology 73 (4): 474–481. https://doi.org/10.1023/B:MICI.0000036994.21650.3a
- Mašínová, T., Bahnmann, B.D., Větrovský, T., Tomšovský, M., Merunková, K. & Baldrian, P. (2017) Drivers of yeast community composition in the litter and soil of a temperate forest. FEMS Microbiology Ecology 93 (2): 223. https://doi.org/10.1093/femsec/fiw223
- Mašínová, T., Yurkov, A. & Baldrian, P. (2018) Forest soil yeasts: decomposition potential and the utilization of carbon sources. Fungal Ecology 34: 10–19. https://doi.org/10.1016/j.funeco.2018.03.005
- Miller, M.A., Pfeiffer, W. & Schwartz, T. (2010) Creating the CIPRES Science Gateway for inference of large phylogenetic trees. In: 2010 Gateway Computing Environments Workshop (GCE), New Orleans USA. IEEE, pp. 1–8. https://doi.org/10.1109/GCE.2010.5676129
- Nguyen, L.T., Schmidt, H.A., Von Haeseler, A. & Minh, B.Q. (2015) IQ-TREE: a fast and effective stochastic algorithm for estimating maximum-likelihood phylogenies. Molecular Biology and Evolution 32 (1): 268–274. https://doi.org/10.1093/molbev/msu300
- Niego, A.G.T., Rapior, S., Thongklang, N., Raspé, O., Hyde, K.D. & Mortimer, P. (2023) Reviewing the contributions of macrofungi to forest ecosystem processes and services. Fungal Biology Reviews 44: 100294. https://doi.org/10.1016/j.fbr.2022.11.002
- Péter, G., Takashima, M. & Čadež, N. (2017) Yeast habitats: different but global. In: Buzzini, P., Lachance, M.A. & Yurkov, A. (Eds.) Yeasts in Natural Ecosystems: Ecology. Springer, Cham, pp. 39–71. https://doi.org/10.1007/978-3-319-61575-2_2
- Rambaut, A. & Drummond, A. (2012) FigTree: Tree figure drawing tool, v1.4.2. Institute of Evolutionary Biology, University of Edinburgh.
- Rao, R.S., Bhadra, B. & Shivaji, S. (2008) Isolation and characterization of ethanol‐producing yeasts from fruits and tree barks. Letters In Applied Microbiology 47 (1): 19–24. https://doi.org/10.1111/j.1472-765X.2008.02380.x
- Ronquist, F. & Huelsenbeck, J.P. (2003) MrBayes 3: Bayesian phylogenetic inference under mixed models. Bioinformatics 19: 1572–1574. https://doi.org/10.1093/bioinformatics/btg180
- Samarasinghe, H., Lu, Y., Aljohani, R., Al-Amad, A., Yoell, H. & Xu, J. (2021) Global patterns in culturable soil yeast diversity. IScience 24 (10). https://doi.org/10.1016/j.isci.2021.103098
- Sampaio, A., Cortes, R. & Leão, C. (2001) Invertebrate and microbial colonisation in native and exotic leaf litter species in a mountain stream. International Review of Hydrobiology 86 (4–5): 527–540. https://doi.org/10.1002/1522-2632(200107)86:4/5<527::AID-IROH527>3.0.CO;2-D
- Sampaio, A., Cortes, R. & Leão, C. (2004) Yeast and macroinvertebrate communities associated with leaf litter decomposition in a second order stream. International Review of Hydrobiology 89 (5–6): 453–466. https://doi.org/10.1002/iroh.200410764
- Sampaio, A., Sampaio, J.P. & Leão, C. (2007) Dynamics of yeast populations recovered from decaying leaves in a nonpolluted stream: a 2-year study on the effects of leaf litter type and decomposition time. FEMS Yeast Research 7 (4): 595–603. https://doi.org/10.1111/j.1567-1364.2007.00218.x
- Solis, M.J.L., Yurkov, A., dela Cruz, T.E. & Unterseher, M. (2015) Leaf-inhabiting endophytic yeasts are abundant but unevenly distributed in three Ficus species from botanical garden greenhouses in Germany. Mycological Progress 14 (1): 1019. https://doi.org/10.1007/s11557-014-1019-6
- Solis, M.J.L., Cruz, T.E.D., Schnittler, M. & Unterseher, M. (2016) The diverse community of leaf-inhabiting fungal endophytes from Philippine natural forests reflects phylogenetic patterns of their host plant species Ficus benjamina, F. elastica and F. religiosa. Mycoscience 57 (2): 96–106. https://doi.org/10.1016/j.myc.2015.10.002
- Štursová, M., Žifčáková, L., Leigh, M.B., Burgess, R. & Baldrian, P. (2012) Cellulose utilization in forest litter and soil: identification of bacterial and fungal decomposers. FEMS Microbiology Ecology 80 (3): 735–746. https://doi.org/10.1111/j.1574-6941.2012.01343.x
- Tennakoon, D.S., Gentekaki, E., Jeewon, R., Kuo, C.H. & Hyde, K.D. (2021) Life in leaf litter: fungal community succession during decomposition. Mycosphere 12 (1): 406–429. https://doi.org/10.5943/mycosphere/12/1/5
- Tennakoon, D.S., Kuo, C.H., Purahong, W., Gentekaki, E., Pumas, C., Promputtha, I. & Hyde, K.D. (2022) Fungal community succession on decomposing leaf litter across five phylogenetically related tree species in a subtropical forest. Fungal Diversity 115 (1): 73–103. https://doi.org/10.1007/s13225-022-00508-x
- Vaughan-Martini, A., Lachance, M.A. & Kurtzman, C.P. (2011) Kazachstania Zubkova (1971). In: Kurtzman, C.P. & Fell, J.W. (5th Ed.) The Yeasts. Elsevier, Amsterdam, pp. 439–470. https://doi.org/10.1016/B978-0-444-52149-1.00034-3
- Voříšková, J. & Baldrian, P. (2013) Fungal community on decomposing leaf litter undergoes rapid successional changes. The ISME Journal 7 (3): 477–486. https://doi.org/10.1038/ismej.2012.116
- Vu, D., Groenewald, M., Szöke, S., Cardinali, G., Eberhardt, U., Stielow, B., De Vries, M., Verkleij, G.J.M., Crous, P.W., Boekhout, T.J.S.I.M. & Robert, V. (2016) DNA barcoding analysis of more than 9 000 yeast isolates contributes to quantitative thresholds for yeast species and genera delimitation. Studies in Mycology 85 (1): 91–105. https://doi.org/10.1016/j.simyco.2016.11.007
- White, T.J., Bruns, T., Lee, S.J.W.T. & Taylor, J. (1990) Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. PCR Protocols: a guide to methods and applications 18: 315–322. https://doi.org/10.1016/B978-0-12-372180-8.50042-1
- Wu, Z.W. & Bai, F.Y. (2005) Kazachstania aquatica sp. nov. and Kazachstania solicola sp. nov., novel ascomycetous yeast species. International Journal of Systematic and Evolutionary Microbiology 55 (5): 2219–2224. https://doi.org/10.1099/ijs.0.63675-0
- Wuczkowski, M., Metzger, E., Sterflinger, K. & Prillinger, H. (2005) Diversity of yeasts isolated from litter and soil of different natural forest sites in Austria. Die Bodenkultur 56 (1/4): 201.
- Yurkov, A. (2017) Yeasts in forest soils. In: Buzzini, P., Lachance, M.A. & Yurkov, A. (Eds.) Yeasts in Natural Ecosystems: Diversity. Springer, Cham, pp. 87–116. https://doi.org/10.1007/978-3-319-62683-3_3