Abstract
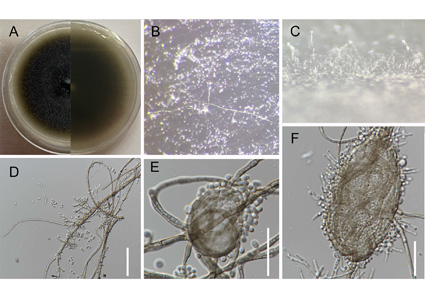
An endophytic fungal species of Exophiala was isolated from the fruits of Amomum tsao-ko during an investigation of the fungal diversity associated with A. tsao-ko in Honghe Prefecture, Yunnan Province, China. Morphological characteristics and the combined internal transcribed spacer (ITS), large subunit nuclear rDNA (LSU), and small subunit nuclear ribosomal rDNA (SSU) loci phylogenetic analysis confirmed the new isolate is a new species, Exophiala hongheensis. Phylogenetically, E. hongheensis is related to E. lavatrina, E. lecanii-corni, and E. mali; however, it can be distinguished from them by its smaller conidia and conidiogenous cells on the lateral side of the mycelium. Descriptions, illustrations, and phylogenetic analysis results of E. hongheensis are provided herein.
References
- Addy, H.D., Piercey, M.M. & Currah, R.S. (2005) Microfungal endophytes in roots. Canadian Journal of Botany 83 (1): 1–13. https://doi.org/10.1139/b04-171
- Alves, A., Crous, P.W., Correia, A.C.M. & Phillips, A.J.L. (2008) Morphological and molecular data reveal cryptic species in Lasiodiplodia theobromae. Fungal Diversity 28: 1–13.
- Beneke, E.S. (1977) Dematiaceous fungi in laboratory-housed frogs. Proceedings of the IV International Conference on Mycoses: ‘The black and white yeasts’. Pan American Health Organization Scientific Publication 356: 101–108.
- Blasi, B., Poyntner, C., Rudavsky, T., Prenafeta-Boldú, F.X., De Hoog, S., Tafer, H. & Sterflinger, K. (2016) Pathogenic yet environmentally friendly? Black fungal candidates for bioremediation of pollutants. Geomicrobiology Journal 33 (3–4): 308–317. https://doi.org/10.1080/01490451.2015.1052118
- Borman, A.M., Fraser, M., Szekely, A., Larcombe, D.E. & Johnson, E.M. (2017) Rapid identification of clinically relevant members of the genus Exophiala by matrix-assisted laser desorption ionization–time of flight mass spectrometry and description of two novel species, Exophiala campbellii, and Exophiala lavatrina. Journal of Clinical Microbiology 55 (4): 1162–1176. https://doi.org/10.1128/jcm.02459-16
- Capella-Gutiérrez, S., Silla-Martínez, J.M. & Gabaldón, T. (2009) TrimAl: a tool for automated alignment trimming in large-scale phylogenetic analyses. Bioinformatics 25 (15): 1972–1973. https://doi.org/10.1093/bioinformatics/btp348
- Carmichael, J.W. (1966) Cerebral mycetoma of trout due to a Phialophora-like fungus. Sabouraudia 5 (1): 120–123. https://doi.org/10.1080/00362176785190211
- Chen, Y. (2007) Caoguo-Baoqing Bencao Zhezhong. In: Zheng, J. (Ed.) Three issues of rare bencao in Nan Song Dynasty [Lexical characteristics of Chinese language]. People’s Medical Publishing House, pp. 588.
- Crous, P.W., Cowan, D.A., Maggs-Kölling, G., Yilmaz, N., Larsson, E., Angelini, C., Brandrud, T.E., Dearnaley, J.D.W., Dima, B., Dovana, F., Fechner, N., García, D., Gené, J., Halling, R.E., Houbraken, J., Leonard, P., Luangsa-ard, J.J., Noisripoom, W., Rea-Ireland, A.E., Ševčíková, H., Smyth, C.W., Vizzini, A., Adam, J.D., Adams, G.C., Alexandrova, A.V., Alizadeh, A., Duarte, E.Á., Andjic, V., Antonín, V., Arenas, F., Assabgui, R., Ballarà, J., Banwell, A,, Berraf-Tebbal, A., Bhatt, V.K., Bonito, G., Botha, W., Burgess, T.I., Caboň, M., Calvert, J., Carvalhais, L.C., Courtecuisse, R., Cullington, P., Davoodian, N., Decock, C.A., Dimitrov, R., Di-Piazza, S., Drenth, A., Dumez, S., Eichmeier, A., Etayo, J., Fernández, I., Fiard, J.P., Fournier, J., Fuentes-Aponte, S., Ghanbary, M.A.T., Ghorbani, G., Giraldo, A., Glushakova, A.M., Gouliamova, D.E., Guarro, J., Halleen, F., Hampe, F., Hernández-Restrepo, M., Iturrieta-González, I., Jeppson, M., Kachalkin, A.V., Karimi, O., Khalid, A.N., Khonsanit, A., Kim, J.I., Kim, K., Kiran, M., Krisai-Greilhuber, I., Kučera, V., Kušan, I., Langenhoven, S.D., Lebel, T., Lebeuf, R., Liimatainen, K., Linde, C., Lindner, D.L., Lombard, L., Mahamedi, A.E., Matočec, N., Maxwell, A., May, T.W., McTaggart, A.R., Meijer, M., Mešić, A., Mileto, A.J., Miller, A.N., Molia, A., Mongkolsamrit, S., Cortés, C.M., Muñoz-Mohedano, J., Morte, A., Morozova, O.V., Mostert, L., Mostowfizadeh-Ghalamfarsa, R., Nagy, L.G., Navarro-Ródenas, A., Örstadius, L., Overton, B.E., Papp, V., Para, R., Peintner, U., Pham, T.H.G., Pordel, A., Pošta, A., Rodríguez, A., Romberg, M., Sandoval-Denis, M., Seifert, K.A., Semwal, K.C., Sewall, B.J., Shivas, R.G., Slovák, M., Smith, K., Spetik, M., Spies, C.F.J., Syme, K., Tasanathai, K., Thorn, R.G., Tkalčec, Z., Tomashevskaya, M.A., Torres-Garcia, D., Ullah, Z., Visagie, C.M., Voitk, A., Winton, L.M. & Groenewald, J.Z. (2020) Fungal Planet description sheets: 1112–1181. Persoonia - Molecular Phylogeny and Evolution of Fungi 45 (1): 251–409. https://doi.org/10.3767/persoonia.2020.45.10
- Glez-Peña, D., Gómez-Blanco, D., Reboiro-Jato, M., Fdez-Riverola, F. & Posada, D. (2010) ALTER: Program-oriented conversion of DNA and protein alignments. Nucleic Acids Research 38 (suppl_2): W14–W18. https://doi.org/10.1093/nar/gkq321
- De Hoog, G.S., Ende, A., Uijthof, J. & Untereiner, W.A. (1995) Nutritional physiology of type isolates of currently accepted species of Exophiala and Phaeococcomyces. Antonie van Leeuwenhoek 68 (1): 43–49. https://doi.org/10.1007/BF00873291
- De Hoog, G.S., Vicente, V., Najafzadeh, M.J., Harrak, M.J., Badali, H. & Seyedmousavi, S. (2011) Waterborne Exophiala species causing disease in cold-blooded animals. Persoonia-Molecular Phylogeny and Evolution of Fungi 27 (1): 46–72. https://doi.org/10.3767/003158511X614258
- De Hoog, G.S., Zeng, J.S., Harrak, M.J. & Sutton, D.A. (2006) Exophiala xenobiotica sp. nov., an opportunistic black yeast inhabiting environments rich in hydrocarbons. Antonie van Leeuwenhoek 90 (3): 257–268. https://doi.org/10.1007/s10482-006-9080-z
- De Hoog, G.S. & Hermanides-Nijhof, E.J. (1997) Rhinocladiella and allied genera. Studies in mycology no.15.
- Gao, J. & Wang, H. (2007) A survey of traditional and modern prescirions containing Caoguo. Tianjin Journal of Traditional Chinese Medicine 24: 15–18.
- Glass, N.L. & Donaldson, G.C. (1995) Development of primer sets designed for use with the PCR to amplify conserved genes from filamentous ascomycetes. Applied and environmental microbiolog 61 (4): 1323–1330. https://doi.org/ 10.1128/aem.61.4.1323-1330.1995.
- Guo, J.W., Yang, L.F., Liu,Y.H., Yang, J., Wang, H.F., Li, L., Liu, Y.H. & Li, W.J. (2016) First report of pseudostem black spot caused by Pestalotiopsis microspora on tsao-ko in Yunnan, China. Plant Disease 100 (5): 1021. https://doi.org/10.1094/PDIS-08-15-0920-PDN
- Hall, T.A. (1999) BioEdit: a user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucleic Acids Symposium Series 41: 95–98.
- He, G., Yang, S. & Wang, Y. (2023) The potential of Amomum tsao-ko as a traditional Chinese medicine: Traditional clinical applications, phytochemistry and pharmacological properties. Arabian Journal of Chemistry 16 (8): 104936. https://doi.org/10.1016/j.arabjc.2023.104936
- Hong, S.S., Lee, J.H., Choi, Y.H., Jeong, W., Ahn, E.K., Lym, S.H. & Oh, J.S. (2015) Amotsaokonal A–C, benzaldehyde, and cycloterpenal from Amomum tsao-ko. Tetrahedron Letters 56 (48): 6681–6684. https://doi.org/10.1016/j.tetlet.2015.10.045
- Hironaga, M., Watanabe, S., Nishimura, K. & Miyaji, M. (1981) Annellated conidiogenous cells in Exophiala dermatitidis, agent of phaeohyphomycosis. Mycologia 73 (6): 1181–1183. https://doi.org/10.2307/3759687
- Ide-Pérez, M.R., Sánchez-Reyes, A., Folch-Mallol, J.L. & Sánchez-Carbente, M.D.R. (2024) Exophiala chapopotensis sp. nov., an extremotolerant black yeast from an oil-polluted soil in Mexico; phylophenetic approach to species hypothesis in the Herpotrichiellaceae family. Plos one 19 (2): e0297232. https://doi.org/10.1371/ journal.pone.0297232
- Jin, H., Yang, Y., Zhang, L., Zhu, X., Wang, L., Ji, P. & Liu, D. (2014) Herbal verification of medicinal Amomum tsao-ko. Medicinal Plant 5 (9): 16.
- Katoh, K., Rozewicki, J. & Yamada, K. (2019) MAFFT online service: Multiple sequence alignment, interactive sequence choice and visualization. Briefings in Bioinformatics 20 (4): 1160–1166. https://doi.org/10.1093/bib/bbx108
- Khan, A.L., Hamayun, M., Ahmad, N., Waqas, M., Kang, S.M., Kim, Y.H. & Lee, I.J. (2011) Exophiala sp. LHL08 reprograms Cucumis sativus to higher growth under abiotic stresses. Physiologia plantarum 143 (4): 329–343. https://doi.org/ 10.1111/j.1399-3054.2011.01508.x
- Lu, H.J., Zhang, Y.X., Liu, W., Liu, Y.L., Li, H. & Sheng, Y.M. (2007) A preliminary study of Phytophthora cactorum on Amomum tsao-ko. Journal of Yunnan Agricultural University 22 (5): 773–775.
- Ma, M., Wang, T. & Lu, B. (2021) Assessment of genetic diversity in Amomum tsao-ko Crevost & Lemarié, an important medicine food homologous crop from Southwest China using SRAP and ISSR markers. Genetic Resources and Crop Evolution 68 (6): 2655–2667. https://doi.org/10.1007/s10722-021-01204-6
- Matsumoto, T., Padhye, A.A. & Ajello, L. (1987) Medical significance of the so-called black yeasts. European Journal of Epidemiology 3 (2): 87. https://doi.org/10.1007/BF00239744
- Maciá-Vicente, J.G., Glynou, K. & Piepenbring, M. (2016) A new species of Exophiala associated with roots. Mycological Progress 15: 1–12. https://doi.org/10.1007/s11557-016-1161-4
- Miller, M.A., Pfeiffer, W. & Schwartz, T. (2012) The CIPRES science gateway: enabling high-impact science for phylogenetics researchers with limited resources. In: Proceedings of the 1st Conference of the Extreme Science and Engineering Discovery Environment: Bridging from the extreme to the campus and beyond. pp. 1–8. https://doi.org/10.1145/2335755.2335836
- Nascimento, M.M., Vicente, V.A., Bittencourt, J.V., Gelinski, J.M.L., Prenafeta-Boldú, F.X., Romero-Güiza, M., Fornari, G., Gomes, R.R., Santos, G.D., Van Den Ende, A.G. & De Hoog, G.S. (2017) Diversity of opportunistic black fungi on babassu coconut shells, a rich source of esters and hydrocarbons. Fungal biology 121 (5): 488–500. https://doi.org/10.1016/j.funbio.2017.01.006
- Nylander, J. (2004) MrModeltest V2. Program Distributed by the Author. Bioinformatics 24: 581–583. https://doi.org/10.1093/bioinformatics/btm388
- Porter, R.B., Gallimore, W.A. & Reese, P.B. (1999) Steroid transformations with Exophiala jeanselmei var. lecanii-corni and Ceratocystis paradoxa. Steroids 64 (11): 770–779. https://doi.org/10.1016/s0039-128x(99)00066-5
- Rambaut, A. (2012) FigTree v1. 4.0. University of Oxford, Oxford, UK.
- Rathnayaka, A.R., Tennakoon, D.S., Jones, G.E., Wanasinghe, D.N., Bhat, D.J., Priyashantha, A.H., Stephenson, S.L., Tibpromma, S. & Karunarathna, S.C. (2024) Significance of precise documentation of hosts and geospatial data of fungal collections, with an emphasis on plant-associated fungi. New Zealand Journal of Botany. [28 pp.] https://doi.org/10.1080/0028825X.2024.2381734
- Rehner, S.A., Samuels, G.J. (1994) Taxonomy and phylogeny of Gliocladium analysed from nuclear large subunit ribosomal DNA sequences. Mycological Research 98: 625–634. https://doi.org/10.1016/S0953-7562(09)80409-7
- Ronquist, F., Huelsenbeck, J.P. (2003) MrBayes 3: Bayesian phylogenetic inference under mixed models. Bioinformatics 19 (12): 1572–1574. https://doi.org/10.1093/bioinformatics/btg180
- Sahu, P.K., Tilgam, J., Mishra, S., Hamid, S., Gupta, A., K, J., Verma, S.K. & Kharwar, R.N. (2022) Surface sterilization for isolation of endophytes: Ensuring what (not) to grow. Journal of Basic Microbiology 62 (6): 647–668. https://doi.org/ 10.1002/jobm.202100462
- Seyedmousavi, S., Badali, H., Chlebicki, A., Zhao, J., Prenafeta-Boldu, F.X. & De Hoog, G.S. (2011) Exophiala sideris, a novel black yeast isolated from environments polluted with toxic alkyl benzenes and arsenic. Fungal biology 115 (10): 1030–1037. https://doi.org/10.1016/j.funbio.2011.06.004
- Stamatakis, A. (2014) RAxML version 8: A tool for phylogenetic analysis and post-analysis of large phylogenies. Bioinformatics (Oxford, England) 30 (9): 1312–1313. https:// doi.org/10.1093/bioinformatics/btu033
- Sudhadham, M., Prakitsin, S., Sivichai, S., Chaiwat, R. & Menken, S.B.J. (2008) The neurotropic black yeast Exophiala dermatitidis has a possible origin in the tropical rain forest. Studies in Mycology 61: 145–155. https://doi.org/10.3114/sim.2008.61.15
- Thitla, T., Kumla, J., Khuna, S., Lumyong, S. & Suwannarach, N. (2022) Species diversity, distribution, and phylogeny of Exophiala with the addition of four new species from Thailand. Journal of Fungi 8 (8): 766. https://doi.org/10.3390/jof8080766
- Vilgalys, R. & Hester, M. (1990) Rapid genetic identification and mapping of enzymatically amplified ribosomal DNA from several Cryptococcus species. Journal of Bacteriology 172: 4238–4246. https://doi.org/10.1128/jb.172.8.4238-4246.1990
- Voigt, K. & Wöstemeyer, J. (2000) Reliable amplification of actin genes facilitates deep-level phylogeny. Microbiological research 155 (3): 179–195. https://doi.org/10.1016/s0944-5013(00)80031-2
- White, T.J., Bruns, T., Lee, S.J.W.T. & Taylor, J. (1990) Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. PCR protocols: a guide to methods and applications 18 (1): 315–322. https://doi.org/10.1016/B978-0-12-372180-8.50042-1
- Woo, P.C., Ngan, A.H., Tsang, C.C., Ling, I.W., Chan, J.F., Leung, S.-Y., Yuen, K.-Y. & Lau, S.K.P. (2013) Clinical spectrum of Exophiala infections and a novel Exophiala species, Exophiala hongkongensis. Journal of Clinical Microbiology 51 (1): 260–267. https://doi.org/10.1128/ JCM.02336-12
- Wu, J.J., Zhu, J.H., Cao, A.C., Yan, D.D., Wang, Q.X., Zhang, C.H. & Li, Y. (2022) Amomum tsao-ko in Yunnan Province: Identification of major pathogenic fungi and screening of control fungicides. Chinese Agricultural Science Bulletin 38 (6): 121. https://doi.org/10.11924/j.issn.1000-6850.casb2021-0369
- Yang, S., Xue, Y., Chen, D. & Wang, Z. (2022) Amomum tsao-ko Crevost & Lemarié: A comprehensive review on traditional uses, botany, phytochemistry, and pharmacology. Phytochemistry Reviews 21 (5): 1487–1521. https://doi.org/10.1007/s11101-021-09793-x
- Yang, X.Q., Feng, M.Y. & Yu, Z.F. (2021) Exophiala pseudooligosperma sp. nov., a novel black yeast from soil in southern China. International Journal of Systematic and Evolutionary Microbiology 71 (11): 005116. https://doi.org/10.1099/ijsem.0.005116
- Yang, Y., Yang, Y., Yan, R.W. & Zhou, G.L. (2010) Cytotoxic, apoptotic, and antioxidant activity of the essential oil of Amomum tsao-ko. Bioresource Technology 101 (11): 4205–4211. https://doi.org/10.1016/j.biortech.2009.12.131
- Yong, L.K., Wiederhold, N.P., Sutton, D.A., Sandoval-Denis, M., Lindner, J.R., Fan, H., Sanders, C. & Guarro, J. (2015) Morphological and molecular characterization of Exophiala polymorpha sp. nov. isolated from sporotrichoid lymphocutaneous lesions in a patient with myasthenia gravis. Journal of clinical microbiology 53 (9): 2816–2822. https://doi.org/10.1128/JCM.00622-15
- Ze, S.Z., Hu, G.H., Ruan, Y.C., Liu, Y.G., Chang, E.F., Nong, S.Z., Lu, M.X. & Li, Y.W. (2011) Pathogenic fungus causing wilt of Amomum tsao-ko. Journal of West China Forestry Science 40 (1): 57–61.
- Zhang, L.Q. & Sheng, L.L. (1991) Preliminary study on Amomum tsao-ko disease. Journal of Yunnan University 13 (3): 255–260.
- Zhang, Y.M., Liu, Y.L., Zhu, K.M., He, Y.L., Wang, Z.Y. & Yang, Z.Q. (2015) Identification of the Pathogen of Amomum tsao-ko Leaf Blast. Journal of West China Forestry Science 44 (1): 149–151. [in Chinese]
- Zhang, Y.X., Lu, H.J., Chen, R.Q., Liang, Z.F. & Liu, Y.L. (2005) Identification of leaf spot of Amomum tsao-ko and trunk canker of Liquidambar for mosana. Journal of Yunnan Agricultural University 20 (3): 438–440.
- Zeng, J.S. & De Hoog, G.S. (2008) Exophiala spinifera and its allies: diagnostics from morphology to DNA barcoding. Medical Mycology 46 (3): 193–208. https://doi.org/10.1080/13693780701799217
- Zong, Y., Ding, M.L., Jia, K.K., Ma, S.T. & Ju, W.Z. (2020) Exploring active compounds of Da-Yuan-Yin in treatment of COVID-19 based on network pharmacology and molecular docking method. Chinese Traditional and Herbal Drugs 51: 836–844.